Memahami perkembangan berbagai tes untuk mendeteksi penyakit COVID-19 perlu diketahui oleh klinisi. Pemeriksaan baku emas untuk penyakit novel corona virus disease (COVID-19) adalah pemeriksaan polymerase chain reaction (PCR). Jenis virus penyebab COVID-19 masuk golongan coronaviridae, serupa dengan virus penyebab SARS (severe acute respiratory syndrome), sehingga penamaannya adalah SARS-CoV-2. Sedangkan istilah COVID-19 secara resmi diperkenalkan WHO pada tanggal 11 Februari 2020. Seluruh dunia saat ini terkena dampak dari penyakit COVID-19, bahkan WHO telah menyatakan keadaan pandemi berlaku untuk seluruh negara tanpa terkecuali.[1-3]
Meskipun banyak aspek biologi yang mempengaruhi tingkat keparahan pada penyakit infeksi tersebut, tetapi penatalaksanaan penyakit yang dilakukan sedini mungkin dapat meningkatkan luaran klinis yang lebih baik. Angka kesembuhan pasien COVID-19 akan semakin meningkat dengan penurunan progresifitas komplikasi sistemik, serta penurunan atau pencegahan terjadinya badai sitokin dan SIRS (systemic inflammatory response syndrome). Untuk itu, dibutuhkan interpretasi tes diagnostik COVID-19 yang tepat sesuai indikasi.[2]
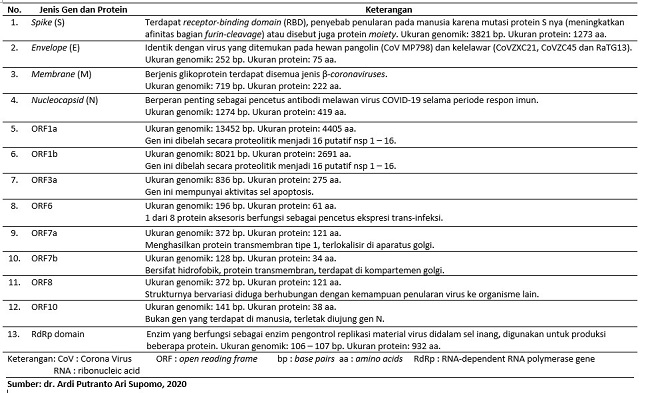
Anatomi Virus SARS-CoV-2
Virus SARS-CoV-2 adalah virus yang mempunyai amplop dengan positive-sense, dimana virus ini bisa langsung bertindak sebagai mRNA (messenger ribonucleic acid) yang berfungsi untuk translasi protein virus saat berada di ribosom sel host. Virus ini memiliki 4 struktur protein utama, selain protein ada juga beberapa jenis gen yang berperan dalam penularan dan penyebaran. Struktur protein dan gen tersebut dapat berperan sebagai skrining, diagnostik, dan monitoring penyakit COVID-19.[2]
Tabel 1. Gen dan Struktur Protein Utama Virus SARS-CoV-2 [2]
Pemeriksaan COVID-19
Jenis pemeriksaan COVID-19 dapat dibedakan menjadi beberapa macam. Berdasarkan tujuan dan karakteristiknya, maka dibedakan menjadi 3 macam, yaitu:
- Pemeriksaan diagnostik, akurasi tinggi tetapi memakan waktu lama
- Pemeriksaan skrining, akurasi rendah tetapi hasil cepat
- Pemeriksaan surveilans, dilakukan pada sekelompok masyarakat dan bertujuan untuk penentuan kebijakan lokal maupun nasional[4]
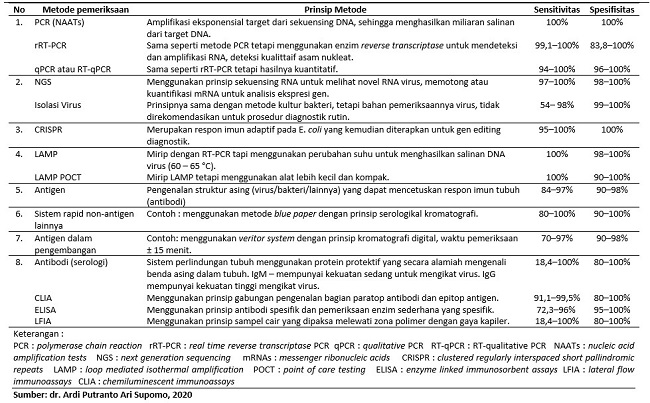
Terdapat beberapa metode pemeriksaan untuk penyakit COVID-19, di antaranya polymerase chain reaction (PCR), next generation sequencing (NGS), clustered regularly interspaced short palindromic repeats (CRISPR), loop mediated isothermal amplification (LAMP), antigen, dan antibodi. Setiap metode pemeriksaan memiliki angka sensitivitas dan sensitivitas dalam mendeteksi infeksi virus SARS-CoV-2. Rangkuman metode, prinsip pemeriksaan, sensitivitas dan sensitivitas dapat dilihat pada tabel 2.[4-8]
Tabel 2. Metode Pemeriksaan COVID-19 [4-8]
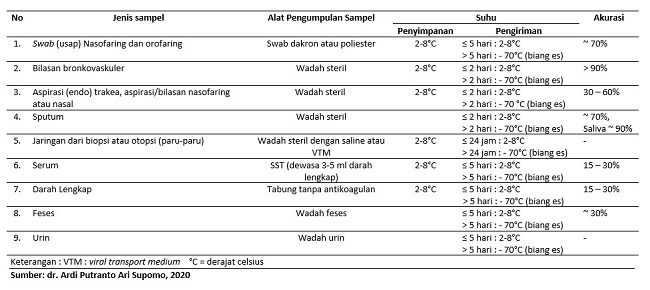
Selain metode pemeriksaan, ada persyaratan lain yang penting diperhatikan untuk mendeteksi COVID-19. Persyaratan tersebut adalah jenis sampel, alat pengumpulan sampel, suhu penyimpanan dan pengiriman, serta ketepatan sampel yang dirangkum pada tabel 3.[3,5]
Tabel 3. Sampel Pemeriksaan COVID-19[3,5]
Interpretasi Pemeriksaan COVID-19
Pemeriksaan COVID-19 dibagi menjadi 2 bagian besar, yaitu pemeriksaan skrining dan pemeriksaan diagnostik. Namun, sebagai pemeriksaan baku emas hingga saat ini masih menggunakan metode PCR.
Interpretasi Pemeriksaan Skrining
Untuk pemeriksaan skrining, biasanya menggunakan sampel darah serum atau darah kapiler. Pemeriksaan ini sering disebut rapid test antibody. Metode menggunakan prinsip pemeriksaan antibodi serologi. Antibodi yang diperiksa ada yang antibodi total, atau antibodi yang terpisah, yaitu immunoglobulin M (IgM), IgA dan IgG. Umumnya interpretasi rapid test antibody menggunakan tanda positif (+) dan negatif (-), atau reaktif dan nonreaktif, tergantung alat tes yang digunakan. Idealnya hasil positif menunjukkan tubuh aktif membentuk antibodi terhadap COVID-19 dan negatif menandakan di dalam tubuh belum terbentuk antibodi, tetapi tes ini memiliki hasil positif palsu yang tinggi.[6-8]
Interpretasi Pemeriksaan Diagnostik
Untuk menegakkan diagnosis, harus menggunakan metode PCR. Sampel yang digunakan adalah cairan bilasan bronkovaskuler, dengan akurasi deteksi berkisar >90%. Sampel swab tenggorok, baik nasofaring atau orofaring, merupakan pengambilan sampling paling aman dan mudah dilakukan dengan akurasi deteksi >70%.[10]
Sebelum mengetahui cara interpretasinya, maka kita harus mengetahui nilai yang dihasilkan dari metode PCR tersebut. Dimana setelah dilakukan pemeriksaan, selanjutnya adalah tahapan validasi yang dilakukan sebelum hasil resmi dikeluarkan. Tahapan ini menggunakan satuan LoD (limit of Detection), yaitu jumlah berapa banyak sampel telah diencerkan (dilusi). Biasanya menggunakan 1 log atau 10x pengenceran. [10]
Rentang atau cut-off nilai Ct (cycle threshold) untuk hasil positif (+) diagnosis COVID-19 berada pada nilai Ct yang mendeteksi adanya salinan gen atau protein virus. Contoh nilai Ct untuk hasil positif COVID-19 adalah nilai Ct ≤38. Sedangkan cut-off nilai Ct untuk hasil negatif (-) diagnosis COVID-19 berada pada nilai Ct yang tidak lagi mendeteksi adanya salinan gen atau protein virus. Contoh nilai Ct untuk hasil negatif COVID-19 adalah nilai Ct ≥40. Nilai Ct ini berbeda-beda pada tiap vendor alat PCR.[10]
Tidak semua jenis gen maupun protein dari virus COVID-19 yang tertangkap oleh PCR. Sebuah studi analisis mendapatkan beberapa gen yang secara signifikan dapat menentukan keadaan klinis pasien, di antaranya:
- Gen ORF1ab, dari sampel nasal dan swab faring dapat menentukan tingkat mortalitas
- Gen N dan ORF1b, dari sampel sputum dapat menentukan progresivitas penyakit
- Gen N dan ORF1ab, dari sampel swab tenggorok dapat menentukan tingkat keparahan penyakit
- Gen E dan RdRP, dari swab nasofaring juga dapat menentukan tingkat keparahan penyakit[10]
Interpretasi Zona Indeterminate dan Positif Palsu
Beberapa jenis alat PCR menyertakan zona indeterminate, dimana rentang nilainya terdapat pada nilai Ct positif dan negatif. Sebagai contoh, pada pusat penelitian kesehatan di Ontario Kanada didapatkan nilai Ct antara 38,1–39,9. Zona indeterminate bisa disebabkan oleh beberapa hal, yaitu kuantitas target virus yang sedikit atau merupakan representasi dari reaktivitas yang nonspesifik (sinyal palsu) pada sampel klinik.[9-11]
Apabila gejala klinis dari pasien kuat mengarah pada infeksi COVID-19, sebaiknya dilakukan pemeriksaan lanjutan dengan target gen yang berbeda, dan pemeriksaan RT-PCR yang tervalidasi sebanding atau lebih sensitif dari pemeriksaan semula. Sebagai alternatif dapat dilakukan sekuensing asam nukleat dengan target gen yang sudah di amplifikasi.[9-11]
Selain zona indeterminate, hasil PCR masih dapat memberikan hasil positif palsu, tetapi persentasenya sangat jarang yaitu <0,01%. Penyebab dari hasil positif palsu dapat dikategorikan menjadi tiga, yaitu:
- Kesalahan pra-analitik: kesalahan yang terjadi sebelum pemeriksaan sampel dilakukan, contohnya kesalahan pelabelan sampel, dan sampel yang terkontaminasi saat pengambilan, pengiriman, atau pemindahan ke aliquot (wadah khusus sampel)
- Kesalahan analitik: kesalahan yang terjadi saat pemeriksaan sampel dilakukan, contohnya kontaminasi reagen alat dan kontaminasi saat tindakan pipetting
- Kesalahan pasca analitik: kesalahan yang terjadi setelah pemeriksaan sampel dilakukan hingga hasil sampai ke pasien, contohnya kesalahan interpretasi kurva amplifikasi dan transkripsi dari hasil[9-11]
Kesimpulan
Virus penyebab COVID-19 mempunyai tingkat penularan yang cukup tinggi. Kekhasan dari virus ini adalah banyaknya gen yang terlibat dalam proses infeksi antar hewan ke manusia, dan manusia ke manusia. Beberapa protein dan gen yang sering digunakan untuk pemeriksaan diagnostik di antaranya adalah gen N, RdRP, ORF1ab dan gen E. Sampel yang digunakan untuk pemeriksaan bervariasi, dari saluran nafas, feses, darah, dan urin. Namun, sampel terbaik adalah menggunakan bilasan bronkovaskuler, saliva, serta swab orofaring atau nasofaring.
Keahlian klinisi dalam pemeriksaan pasien COVID-19 dan/atau orang yang diduga pasien COVID-19 sangat dibutuhkan di masa pandemi ini, termasuk keahlian interpretasi hasil tes diagnostik. Metode pemeriksaan COVID-19 dibagi menjadi skrining antibodi dengan metode serologi (CLIA, ELISA atau ELFA), dan diagnostik antigen dengan metode RT-PCR. Metode serologi menggunakan hasil (+) dan (-), sedangkan metode RT-PCR menggunakan nilai Ct. Pada metode RT-PCR terdapat zona indeterminate yang berpotensi menghasilkan hasil positif palsu.
Selain pemeriksaan laboratorium, pemeriksaan penunjang lain yang digunakan sebagai konfirmasi infeksi COVID-19 adalah pemeriksaan radiologi CT-scan toraks, juga pemeriksaan hematologi rutin dan hitung jenis leukosit.. Data dari hasil laboratorium dan radiologi dapat digunakan sebagai skrining, diagnostik, dan monitoring dari pasien terinfeksi COVID-19. [12]